Research
team (from left to right): Dr Jing Xiang (Research Fellow); Mr Yau-Fu
Chan (Research Assistant); Prof T C Lau (Project Coordinator); Mr
Ru-Wei Wang (Research Assistant)
The amount of organic
pollutants in wastewaters discharged by various industries is
increasing every year and is causing serious global environmental
problems. These pollutants include a wide range of persistent organic
chemicals, such as pharmaceuticals and endocrine-disrupting compounds.
We are now faced with the challenge of removing these compounds from
their effluents before they are discharged. In this regard,
photocatalysis is a promising technology for wastewater treatment as it
offers many advantages over conventional and other advanced treatment
options. Traditional physical techniques (adsorption on activated
carbon, ultrafiltration, reverse osmosis, coagulation by chemical
agents, ion exchange on synthetic adsorbent resins, etc.) have been
used for the removal of these pollutants, but these methods only
succeed in transferring organic compounds from water to another phase,
thus creating secondary pollution. Microbiological or enzymatic
decomposition, biodegradation, ozonation and advanced oxidation
processes such as Fenton (Fe2+ + H2O2), photo-Fenton (Fe2+ + H2O2 +
UV), H2O2/UV processes have also been used for organic pollutants
removal from wastewaters, but these methods are either ineffective or
too expensive. On the other hand, photocatalysis is energy saving and
is able to completely oxidize the organic pollutants to water and
carbon dioxide. Metal oxide semiconductor nanoparticles have been used
as high activity photocatalysts; such as ZnO, TiO2, CdS, Fe2O3 and
HNbO3. These semiconductors, however, can only be excited by UV light
due to their large band
|
|
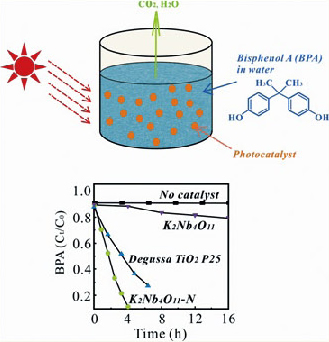
Photodegradation of bisphenol A in water (an emerging organic pollutant)
gaps. For better utilization of sunlight and indoor
illumination, it is desirable to develop photocatalysts that can be
excited by visible light. In this project we have developed a series of
highly efficient visible light photocatalysts based on K2Nb4O11 and
related niobium oxides. These semiconductors are doped with various
metals and non-metals. Doping with metals can lead to increased
photocatalytic efficiencies, while doping with non-metals can shift the optical absorption of the photocatalysts to the visible
region. We have successfully prepared a series of doped niobium oxides
which show high photocatalytic activity in the degradation of organic
pollutants such as Orange G (an organic dye) and Bisphenol A (an
endocrine disruptor) under visible light (λ > 400 nm).
For example, the photocatalytic activity of N-doped
K2Nb4O11 is much higher than that of undoped K2Nb4O11 and TiO2 P25,
indicating the great positive effect of nitrogen doping. Another
advantage of doped K2Nb4O11 is that they readily settle into the bottom
of the solution after photocatalysis, and can be readily recovered and
reused without loss of photoactivity. On the other hand, TiO2 cannot be separated from the solution after photo-catalysis even by
centrifugation and therefore it is difficult to be reused. Our project
should lead to the development of a novel and practical nanotechnology
for environmental remediation.
Prof Tai-chu LAU
Department of Biology and Chemistry
City University of Hong Kong
bhtclau@cityu.edu.hk
|
