 Principle Investigator Dr. CHENG Shuk Han, Department of Biology and Chemistry, City University of Hong Kong
Principle Investigator Dr. CHENG Shuk Han, Department of Biology and Chemistry, City University of Hong Kong
To grow and spread, tumors need blood vessels to provide
nutrients. Scientists are trying to find ways to stop angiogenesis.
Angiogenesis inhibitors stimulate cells to stop the
angiogenes is
process. Hence, angiogenesis inhibition therapy can help
chemotherapy drugs and radiation therapy work more effectively when
given in combination. Vascular integrins, in particular
alpha-v-beta-3, are important regulators of angiogenesis, including tumor angiogenesis. The integrin alpha-v-beta-3
is highly expressed on activated endothelial cells and tumor cells but is
not present in resting endothelial cells and most normal organ
systems. The cyclic arginine-glycine-aspartic acid (RGD) peptide recognizes
alpha-v-beta-3 integrin receptors and thus target to cancer cells.
Dr. Cheng’s research team has shown that nanomaterials,
such as carbon nanotubes, can be used as promising drug delivery
vesicles. They have developed a new type of nanotherapeutics, namely multifunctional
carbon nanotubes, to target and destroy aggressive ectopic angiogenesis.
The generated nanoconjugates mainly accumulated in the
blastoderm cells which facilities the drug delivery applications.
To generate the anti-angiogenic agents, they have attached
anti-angiogenic drug, together with targeting moieties and
tracking dye, onto the same carbon nanotubes. The targeting
molecules, RGD, can target newly formed blood vessels
while the anti-angiogenic drugs Thalidomide can inhibit the
angiogenesis process. The novel nanotherapeutic agents
can prevent drugs from interacting with normal cells, thus
avoiding side effects.
The zebrafish system is an important model for studying human
development and diseases. Transparency of zebrafish embryos facilitates
visual inspection of cardiovascular system as well as of changes in blood
vessel growth. Previous research has also shown that drugs causing
anti-angiogenesis in human have similar effect in the developing
zebrafish embryo. Dr. Cheng’s research team have successfully applied
the zebrafish system to screen for angiogenic modulating activities in
Chinese herbs and derived compounds.
|
|
The biodistribution, efficacy and
biocompatibility of the generated nanotherapeutics were tested in the
transparent zebrafish embryos. The delivery vector carbon nanotubes showed good
biocompatibility in the loaded developing zebrafish embryos, and no toxic effects
were observed in the whole life cycle when they were loaded into
the embryos just after fertilization. Carbon nanotubes conjugated with
RGD illustrated efficient angiogenesis targeting and further mammalian
cell xenograft assay demonstrated that the developed anti-angiogenic
nanotherapeutics specifically inhibited the ectopic angiogenesis in
zebrafish embryos.
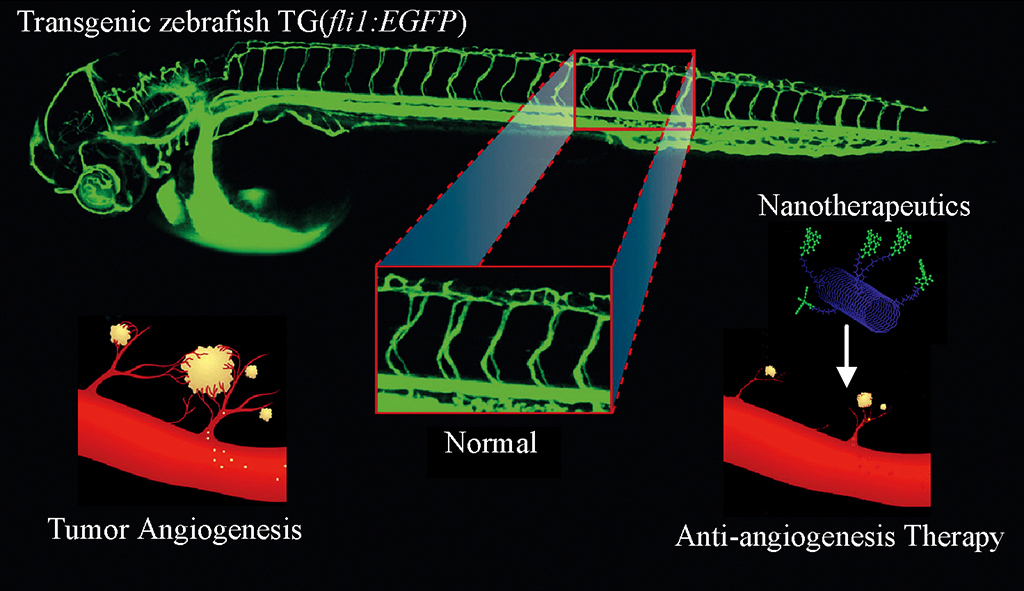
The blood vessels of transgenic zebrafish
embryos can be easily visualized under confocal microscope. While ectopic
angiogenesis is a necessary for tumor growth and expansion, multifunctional
nanotherapeutics was designed to target and destroy tumor angiogenesis specifically
and simultaneously.
This study attempts to capitalize on the highly
reactive nature of the carbon nanotubes and conjugate several moieties onto the
surface to achieve targeted delivery of small dosages of anti-angiogenic drugs to newly
formed blood vessels. This study generated nanosized tools which can be used for specific
and simultaneous delivery of treatment to tumor angiogenic sites. This project forms an
essential part in our current drive to apply nanotechnology to improve human health.
Dr Shuk-han CHENG
Department of Biology and Chemistry
City University of Hong Kong
bhcheng@cityu.edu.hk
|
