This project aimed to develop a new non-invasive prenatal testing technology for providing genetic and chromosomal information
about a fetus in a safe manner. The project team (Fig. 1) had successfully developed such a technology in the first phase of the project
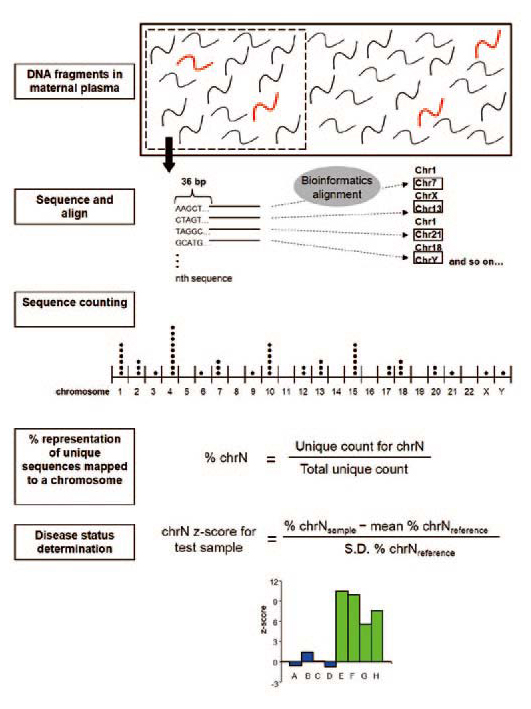
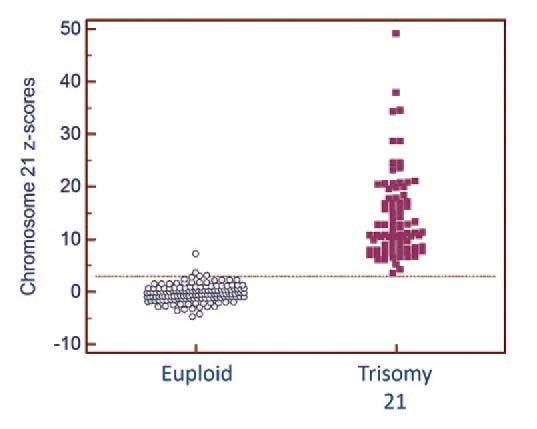
(Fig. 2) and then went on to validate it for Down syndrome testing in the second phase (Fig. 3). The resulting test is over 99% accurate.
In addition to testing for Down syndrome, the project team has also extended this technology to test other chromosomal abnormalities
(such as trisomy 18 and trisomy 13), as well as a number of single gene disorders, e.g. beta-thalassaemia. As an exercise to push the
technology to its limit, the project team was the first to show that the entire fetal genome could be sequenced from maternal plasma.
This work was published as a cover article in Science Translational Medicine and was widely publicised in international media.
The project team has created a valuable intellectual property portfolio. Part of this portfolio has been licensed to Sequenom and Illumina,
two international biotechnology companies, as well as Xcelom. The latter is a local biotechnology startup company based in the Hong Kong
Science Park founded by three members of the project team. A number of previous students of the project team now work at Xcelom.
Commercial tests based on this technology have been available since 2011 and have now been performed in over 90 countries and used
by millions of pregnant women. This technology has received endorsements from many international professional bodies. The use of this
technology has resulted in an approximately 30%-50% reduction in the use of invasive tests, hence resulting in a paradigm shift in prenatal
medicine. Members of the project team have also received multiple
international awards and honours based on this work, including
election to the Royal Society, the US National Academy of Sciences
as well as the King Faisal International Prize in Medicine in 2014. |
|
|
|
|
|
|
| |
 |
|
|
|
Fig 2. Sequencing of maternal plasma DNA for the
non-invasive prenatal detection of fetal chromosomal
disorders |
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
 |
|
|
|
Fig 3. High accuracy for detecting Down syndrome
prenatally |
|
|
|
|
|
|
|
|
Prof Yuk Ming Dennis LO
Department of Chemical Pathology
The Chinese University of Hong Kong
loym@cuhk.edu.hk
|
