Oxygen is a fundamental requirement for the survival
of metazoan organisms. The deprivation of oxygen (or hypoxia) is critical in the pathogenesis of major
diseases such as stroke, myocardial infarction, cancer, impaired tissue regeneration, and reperfusion injury
following organ transplantation. Hypoxia can also be part of the normal
physiology, for example, during embryonic development. The central pathway for
sensing oxygen fluctuations has been recently identified. The transcription factor hypoxia inducible factor 1
(HIF-1) has been shown to play essential role in the maintenance of oxygen homeostasis in cells.
HIF-1 is a heterodimer composed of an oxygen regulated HIF-1α subunit and a
constitutively expressed HIF-1β subunit. HIF-1α levels are controlled by regulated
proteolysis through an oxygen sensitive mechanism. Under normoxic conditions,
HIF-1α undergoes prolyl hydroxylation and is ligated by von Hippel-Lindau protein (pVHL), an E3 ubiquitin
ligase and then degraded by the proteosome. Under hypoxia, prolyl hydroxylation is inhibited, HIF-1α then
accumulates in the cytoplasm and translocates into the nucleus where it forms a dimer with the HIF-1β
subunit. The dimer then complexes with coactivator p300 and transactivates HIF responsive genes. Within
any given cell type, HIF-1α regulates the expression of hundreds of target genes that involves in
angiogenesis, glucose metabolism, erythropoiesis, cell proliferation and
differentiation.
Although the cellular responses to hypoxia are relatively well known, at molecular level, how changes of oxygen concentrations are translated into cellular signals remains largely unclear particularly in the in vivo contexts. During organogenesis or tissue regeneration hypoxia influences proliferation and differentiation of stem/progenitor cell populations. The hypoxia/HIF pathway is considered as a potent signaling pathway that affects the fundamental properties of stem cells including embryonic stem cells, induced pluripotent stem cells, and various adult stem cells.
|
|
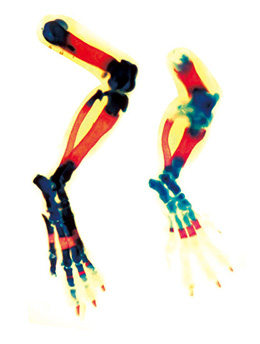
Using skeletal tissue specific gene knockout
strategy we found that bone cells (osteoblasts) restricted overexpression or deletion of
HIF-1α produces marked changes in the vascularization and formation of long bones
but not in the flat bones of the skull. These observations suggest a different relationship
between angiogenesis and osteogenesis at this skeletal site. We further analyzed the role
HIF-1α in the development of condensing mesenchyme using in vitro
and in vivo methodologies including mesenchymal stem cells (MSC) colony forming unit assay, cytochemistry, histology,
immunostaining, in situ hybridization, and radiographic evaluation, et al. We found
that mice lacking HIF-1α in MSCs had smaller and less mineralized
bones than their control littermates, with disorganized mesenchymal
condensation during early stage of development. Deletion of HIF-1α impaired MSCs self-renewal and osteoblast differentiation and maturation. This is achieved at least partially through
regulating MSCs cell cycle and transcriptional control of osteoblast specific transcription
factor osterix by HIF-1α.
We next determined the role of HIF-1α in tissue regeneration genetically and
pharmacologically using skeletal repair models. The results showed that the
stem/progenitor cells were located in a hypoxic microenvironment during
regeneration. Targeting the hypoxia/HIF pathway using small molecules could serve as a therapy for promoting MSCs expansion and
skeletal regeneration. The results of this project has provided new scientific knowledge about
the role of HIF-1α in regulating adult stem cell self-renewal and differentiation, which
might facilitate the discovery of novel therapies for tissue or organ
regeneration.

Dr Chao Wan (third from the left) and his research team
Dr Chao WAN
School of Biomedical Sciences
The Chinese University of Hong Kong
cwan@cuhk.edu.hk
|
